In 2017, with the release of the “Opinions on Deepening the Reform of Examination and Approval System and Encouraging Innovation in Pharmaceutical Medical Devices†and the gradual implementation of the priority review and approval system, the signals encouraging the innovation of pharmaceutical companies are continuously released; China officially joined ICH, and generic drugs are consistent. Sexual evaluation is effective at the beginning... These are undoubtedly bringing companies closer to international standards and towards innovation, which is also a wave of domestic pharmaceutical industry. In this wave, it will be a devastating disaster for small and medium-sized enterprises with insufficient innovation, and it will be a icing on the cake for enterprises that regard innovation as their core competitiveness. In 2017, many innovative companies focused on new drug research and development received considerable financing.
28 non-listed innovative R&D pharmaceutical companies in 2017
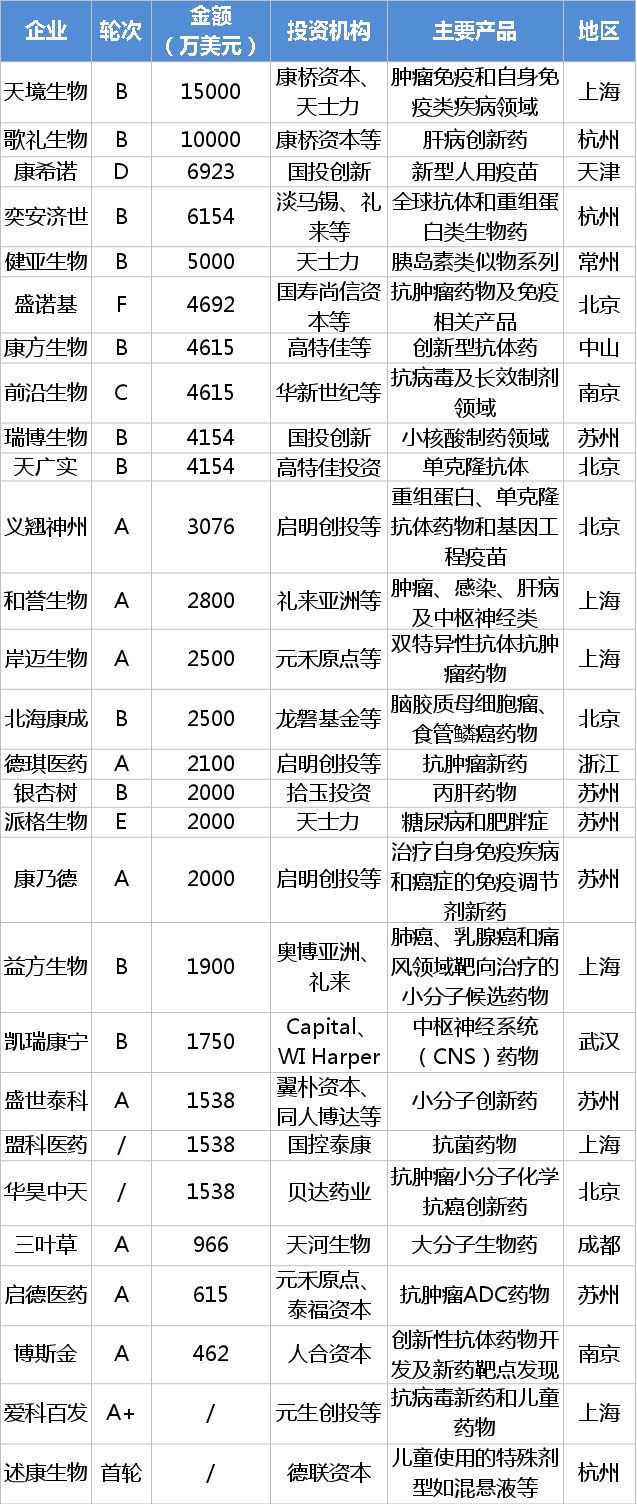
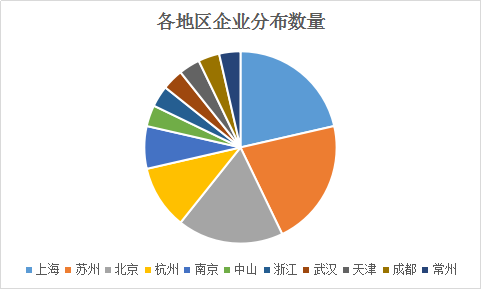
From the above table, there are more companies in the A and B rounds of financing. We can boldly speculate that investors prefer the enterprises from the transition to the transition from maturity to maturity. Qiming Venture Capital, SDIC Innovation, and Lilly are still major investment institutions. There are still many pharmaceutical companies such as Tasly, Beida Pharmaceutical and Tianhe Bio in the investment camp. From the perspective of geographical distribution, there are more Shanghai, Beijing and Suzhou. The specific distribution map is as follows:
Tianjing Biology, Kang Xinuo, and Songli Biology, which have more financing, have their own characteristics:
Tianjing Bio is optimistic about focusing on biopharmaceuticals, wide product layout, and rapid advancement. According to Tianjing Bio, the company has 12 biopharmaceutical projects under development, including 7 research and fusion protein drugs, and 5 indications include diabetes, tumors, autoimmune diseases and other drugs. It is expected that by 2019, 2-3 new drugs will enter the clinical stage, and at least 7 innovative drugs will be declared or started clinically in the United States and China. At present, the clinical trial application for two imported therapeutic biological products, TJ 101 injection and TJ 301 injection, has been accepted by CDE.
As a biopharmaceutical manufacturer, Connino focuses on the development and production of new human vaccines. The recombinant Ebola virus vaccine launched this year is China's first innovative recombinant vaccine with independent intellectual property rights. It is one of the most influential innovative drugs in 2017. In addition, the clinical application of Kangxi's six products has been contracted by CDE. Among them, ACYW135 group meningococcal conjugate vaccine and lyophilized group A group C meningococcal conjugate vaccine have been approved.
The most optimistic about the songs and creatures is its class 1.1 new drug, Danolive (ASC08), which is the first domestic drug in the field of direct antiviral drugs for hepatitis C. Its application for production has been submitted on 2017-01- 03 was accepted by CDE. In addition, another hepatitis C innovative drug, Revidavir (ASC16), has been approved for clinical trials and has been included in the priority review category. It is believed that the injection of this fund will accelerate the listing of the former and the development of the latter.
In summary, we can see that a wide product layout and possession of heavy products is the key to financing. However, as some companies are in their infancy, there are restrictions on technology, talent and capital. Most companies will concentrate on developing a heavy product. The inflow of financing funds in 2017 will accelerate the development of these heavy products.
Liver cancer Chinese medicine. Drug - acroridine
As a new drug for the treatment of advanced hepatocellular carcinoma, Shengkor's original research product is a modern class 1 traditional Chinese medicine and natural medicine with global independent intellectual property rights. The product has entered the phase III clinical trial stage. In addition, Shenguoji's Class 1 new drug oncolytic virus (Pexa-Vec) is also about to launch Phase III clinical trials in China.
[Kang Fang Bio] monoclonal antibody innovative drug - AK101
AK101 injection (recombinant human anti-IL-12/IL-23 p40 monoclonal antibody injection) was approved in China on September 27, 2017. This product is the first new drug to be declared clinically since the establishment of Kangfang Bio. It is also the first monoclonal antibody drug targeting IL-12/IL-23 p40 in China. In addition, there are two new drugs in the domestic Kangfang Biological - AK104, AK105 injection has been accepted by CDE.
Black and White Ultrasound Scanner
"Black and White Ultrasound Scanner" is to emit ultrasonic waves, so that the ultrasonic waves encounter barriers to generate echoes, use the computer to collect these echoes, convert them into corresponding images and display them on the screen, and the images measured by the ultrasonic probe are black and white, so it is called " Black and White Ultrasound Scanner" is also the earliest diagnostic technology adopted by b-ultrasound.
"Black and White Ultrasound Scanner" has the advantages of non-invasive, high sensitivity, low cost and convenient operation
Black And White Ultrasound Scanner,Animal Ultrasound Scanner,Palm Veterinary Ultrasound Machine,Animal Veterinary Ultrasound Machine
Mianyang United Ultrasound Electronics Co., Ltd , https://www.uniultrasonic.com