Reduce the risk by 71%! Prostate cancer combination therapy phase 3 results released
February 07, 2018 Source: WuXi PharmaTech
Window._bd_share_config={ "common":{ "bdSnsKey":{ },"bdText":"","bdMini":"2","bdMiniList":false,"bdPic":"","bdStyle":" 0","bdSize":"16"},"share":{ }};with(document)0[(getElementsByTagName('head')[0]||body).appendChild(createElement('script')) .src='http://bdimg.share.baidu.com/static/api/js/share.js?v=89860593.js?cdnversion='+~(-new Date()/36e5)];Recently, Astellas Pharma and Pfizer jointly announced the use of XTANDI (enzalutamide) and androgen deprivation therapy (ADT) combination therapy in the treatment of non-metastatic resistance to prostate. Positive effects were obtained in clinical phase 3 trials of patients with castration-resistant prostate cancer (CRPC). The trial results show that this combination therapy can reduce the risk of tumor metastasis or death in patients by 71% compared to single ADT therapy! Detailed data for this trial will be presented at the 2018 Genitourinary Cancers Symposium in San Francisco.

Prostate cancer is the second largest cancer in men worldwide. More than 160,000 men in the United States are expected to be diagnosed with prostate cancer by 2018. In the middle of 2015, the number of new prostate patients in the EU was estimated to be 365,000. CRPC refers to patients whose prostate cancer is still developing further, although the level of androgen has dropped to castration. Patients with tumors that have not metastasized in these patients usually have elevated prostate-specific antigen (PSA) levels. If they have a rapid rise in PSA levels, they are prone to tumor metastasis.
Enzalutamide is a specific androgen receptor inhibitor that blocks not only the binding of androgens to receptors, but also inhibits the transfer of receptors into the nucleus and inhibits the binding of androgen receptors to DNA. Therefore, it is not only an antagonist of the androgen receptor, but also has an inhibitory effect on the androgen signaling pathway. In 2012, the US FDA approved it for the treatment of CRPC patients whose tumors have metastasized.
In a randomized, double-blind, placebo-controlled multinational clinical phase 3 trial called PROSPER, nearly 1,400 patients with untransferred CRPC were treated with enzalutamide + ADT or placebo + ADT. The primary endpoint of the trial was metastasis free survival (MFS). The results of the trial showed that the combination therapy of enzalutamide and ADT significantly prolonged MFS in patients with ADT. The median MFS of combination therapy was 36.6 months, while the median MFS of ADT monotherapy was 14.7 months (n=1401, p <0.0001).
The secondary endpoint of the trial included time to PSA progression, the time to first use of new antineoplastic therapy and overall survival. The trial results showed that combination therapy reduced the risk of elevated PSA levels by 93% (P < 0.0001), and combination therapy delayed the PSA level by an average of 33.3 months compared with ADT monotherapy. At the same time, combination therapy reduced the risk of patients needing to use new anticancer drugs by 79% (p<0.0001).
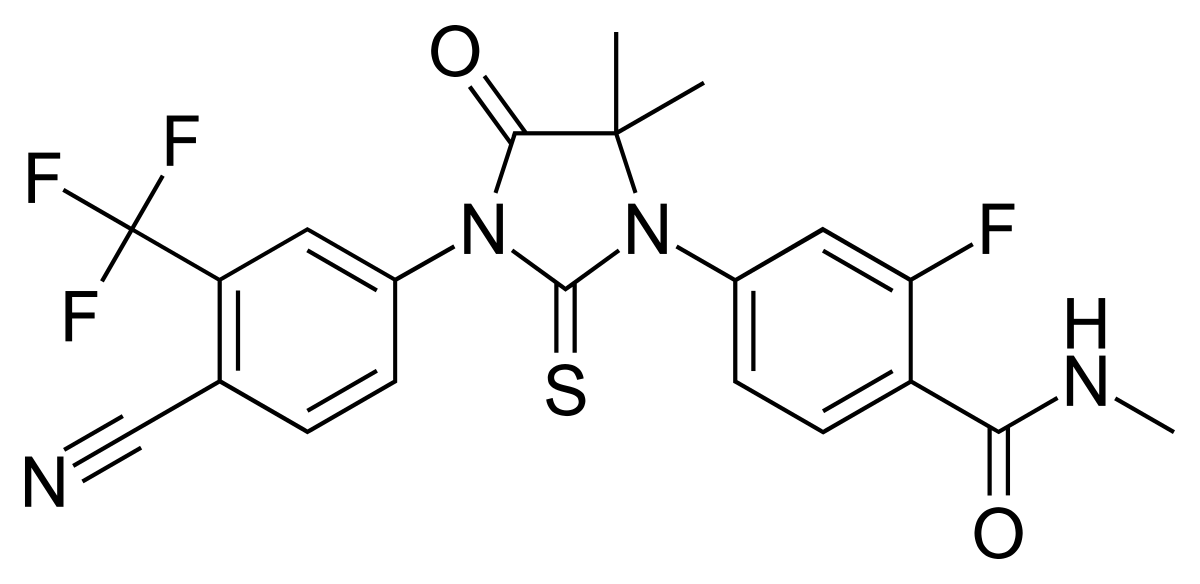
â–² Enzalutamide molecular structure (Source: Wikipedia)
The side effects that appear in the PROSPER clinical trial are consistent with the side effects of enzalutamide in previous clinical trials. Approximately 31% of patients had grade 3 or more side effects after receiving combination therapy, and the value of patients receiving ADT monotherapy was 23%. The most common side effects are high blood pressure and fatigue.
"In the case of CRPC patients whose tumors have not metastasized, the delay in metastasis and the further development of advanced prostate cancer is an important unmet medical need. There are currently no approved systemic therapies in the United States for the treatment of tumor-untransferred CRPC patients. Dr. Maha Hussain of the Robert H. Lurie Comprehensive Cancer Center at Northwestern University said: "In the PROSPER clinical trial, the combination of enzalutamide and ADT can significantly delay the onset of tumor metastasis. If approved, it will give the tumor a non-metastatic CRPC. Patients provide an important new treatment option."
A marketing application based on the PORSPER test results has been submitted to the US FDA and the European Medicines Agency (EMA). These data will then be submitted to regulatory agencies around the world. We look forward to hearing from you soon the good news that this new drug has been approved for the benefit of patients!
Reference materials:
[1] Phase 3 PROSPER Trial Shows XTANDI® (enzalutamide) Significantly Reduced the Risk of Metastasis or Death by 71 Percent in Men with Non-Metastatic Castration-Resistant Prostate Cancer
[2] Evidence for the efficacy of enzalutamide in postchemotherapy metastatic castrate-resistant prostate cancer
Manual Massage Collapsible Foot Spa Massager
The Manual Massage Foldable Foot Spa Massager is a portable compact device designed to give your feet a relaxing massage experience. Its foldable design makes it easy to store and transport, making it ideal for the frequent traveler.
Massagers typically have multiple massage rollers that apply pressure to different areas of the foot, providing a deep-tissue massage that can help relieve tension and improve circulation. Some models also feature heating and vibration to further enhance the massage experience.
To use the massager, you simply fill it with water and turn it on. The water will heat up and the massage rollers will go into action to provide a soothing massage that will help relax your feet.
Overall, the Manual Massage Foldable Foot Spa Massager is an excellent choice for anyone looking to relieve foot pain and tension in the comfort of their own home.
Manual Massage Collapsible Foot Spa Massager,Foot Bath Massager Machine,Electric Heated Foot Spa,Foot Bath Tub
Huaian Mimir Electric Appliance Co., LTD , https://www.mimirfootbath.com