Inducing pluripotent stem cells to treat heart disease, hope or worry?
June 04, 2018 Source: Science and Technology Daily
Window._bd_share_config={ "common":{ "bdSnsKey":{ },"bdText":"","bdMini":"2","bdMiniList":false,"bdPic":"","bdStyle":" 0","bdSize":"16"},"share":{ }};with(document)0[(getElementsByTagName('head')[0]||body).appendChild(createElement('script')) .src='http://bdimg.share.baidu.com/static/api/js/share.js?v=89860593.js?cdnversion='+~(-new Date()/36e5)];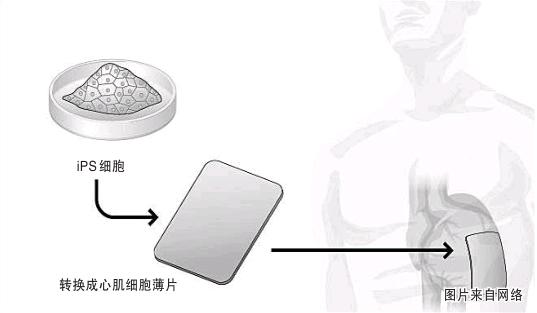
According to a recent report by the British "Nature" magazine, the Ministry of Health, Labor and Welfare (the main department responsible for medical and social security) gave a green light to the team led by Osaka University's cardiac surgeon Ze Fangshu in mid-May to approve the team. A revolutionary reprogramming technique, a clinical research project to induce pluripotent stem cells (iPS cells) for the treatment of heart disease, is the second clinical application of iPS cells.
The research team plans to implement the world's first clinical trial of iPS cells for heart disease next year. The “passport†issued by the Ministry of Health, Labour and Welfare has excited many researchers in the industry, but many people are worried that the current regulatory measures are not sufficient.
iPS cells have great potential
The so-called induced pluripotent stem cells are obtained by reprogramming adult cells in body tissues such as skin and blood into cells similar to the embryo state, thereby regaining the ability to differentiate into various other types of cells.
In 2006, Yamanaka Yamanaka, who is currently the director of the iPS Cell Research Institute at Kyoto University, used a viral vector to transfer a combination of four transcription factors into differentiated somatic cells and reprogram them to obtain a cell type similar to embryonic stem cells. - iPS cells. Yamanaka is called "the father of iPS" and won the Nobel Prize in Physiology or Medicine in 2012.
For regenerative medicine such as repairing damaged organs, iPS cells are a huge boon. Although it is difficult to avoid ethical controversy because it can be used to make germ cells, it faces fewer moral and ethical controversies than real human embryonic stem cells, so it is sought after by many scientists and medical professionals, hoping to try this technology. Treat patients.
On March 28, 2017, the British "Nature" magazine officially reported that a 60-year-old man in Japan became the first person in the world to receive retinal cells produced by other iPS cells. This time, Osaka University will use iPS cells to treat heart disease.
Repair damaged heart
Previously, Zefang Shu and colleagues used iPS cells to create a sheet containing 100 million cardiomyocytes. Studies in pigs have shown that transplanting these 0.1 mm thick, 4 cm long cell sheets onto the heart of pigs can improve their heart function. Ze Fangshu said that the post-implanted cells are not integrated with the heart tissue. The main function of these cardiomyocytes is to release growth factors and help the damaged myocardium to regenerate.
It is reported that in the latest plan, they will use the healthy human iPS cells provided by Kyoto University to culture cardiomyocytes to make disc-shaped myocardial pieces with a diameter of several centimeters and a thickness of about 0.1 mm, and transplant the myocardial pieces to patients. On the heart to improve myocardial function in patients.
Scientists believe that one advantage of cell sheets is that they can create their own cell matrix and maintain structure without the need for stents made of foreign materials. Philip Monnascher, a cardiac surgeon at the Georges Pompidou European Hospital in Paris, was involved in the manufacturing process of tissue sheets. He said: "This is an elegant and intelligent way of delivering cells."
Pharmacologist Wolfram Hubertus Zimmermana of the University of Göttingen Medical Center in Germany is also developing a therapy for treating heart disease with iPS cells. He said that the latest attempt is based on research done by Ze Fangshu and colleagues over the past 15 years.
Zefangshu's clinical trial will begin next year. Three patients will be treated initially, followed by another seven, bringing the entire clinical trial to 10. If this technology proves to be safe and demonstrates some efficacy, then the therapy will be commercially viable based on the “fast track system†in the field of regenerative medicine in Japan. This allows researchers to circumvent expensive large-scale clinical trials designed to demonstrate the efficacy of a treatment, while small-scale trials to demonstrate the safety and efficacy of therapies.
Thomas Eisenhagen, a pharmacologist and chairman of the Cardiovascular Research Center at the University of Hamburg in Germany, said: "This will attract worldwide attention because many groups are working in this direction."
Experts worry about inadequate regulation
Although academics are optimistic about clinical trials, they are worried about their commercialization. The Fast Track System was launched in 2014 to accelerate the use of potential life-saving therapies. Critics say the system is flawed because it allows the person to sell it to the patient without getting enough data to prove that the therapy is effective.
In addition, some researchers question whether the threshold for approval of therapies for commercial use is too low. Even if these cells are safe, there is a risk of any surgery, and patients are likely to abandon other treatments because of the safety and efficacy of less certain or even ineffective treatments. Ethicists and regulators also believe that for any new therapy, its benefits must exceed the risk to be of value.
According to Jing Fangshu, a cardiologist at Kyoto University in Japan, in addition to meeting safety requirements, researchers should also prove that their therapy is effective, which requires testing on more people. He said that the evaluation process should use random, controlled clinical trials, which is the gold standard for validating medical research. “Although iPS cell therapy has great potential, due to the lack of controlled experiments, we are still not sure whether it really works. The biggest problem is that Japan currently lacks a sufficient evaluation system.â€
The Ministry of Health, Labour and Welfare spokesperson did not agree. He told Nature that the current approval criteria are sufficient, because even if commercial applications have been approved, researchers still need to continue to provide evidence that the therapy is effective.
For the current debate, Ze Fangshu agrees with the importance of control experiments to prove efficacy. But he also pointed out that he is still working on the framework of the Japanese rules - he does not need a control experiment before commercialization. The Ministry of Health, Labour and Welfare approved the therapy by acknowledging that the therapy was “scientific and ethical†on the patient, and whether the therapy really worked, “we must find the answer nowâ€. (Reporter Liu Xia)
Medicine Peptides peptides usually regulate the immune function of the body. It also has the effect of increasing the immune barrier of human body, improving cellular immunity and humoral immunity. Cosmetic peptides are indispensable small molecules that link proteins and amino acids. Beauty peptide has many functions in human body and is an indispensable tissue component. Generally speaking, beauty peptide can regulate the nutrition and immunity of human body. In addition, hairdressing peptide still can adjust the metabolism of cholesterol inside human body. For example, when the body is deficient in cholesterol or peptides, the metabolism of cholesterol in the body becomes impaired."
cosmetic peptides list,cosmetic peptides for sale,cosmetic peptide suppliers,buy cosmetic peptide,cosmetic peptide manufacturers
Shaanxi YXchuang Biotechnology Co., Ltd , https://www.peptidenootropics.com