3.15 What are the flight inspection and verification dynamics of CFDA and local provincial food and drug administrations last week? Xiaobian made a summary for everyone, and see below:
CFDA
The General Administration informed that six pharmaceutical companies were received GMP
On March 5, the State Food and Drug Administration issued two notices to announce the situation of six pharmaceutical companies suspected of illegal production of drugs. Recently, the General Administration of the People's Republic of China respectively to Xiyunpeng Pharmaceutical Co., Ltd., Shanxi Huawei Pharmaceutical Co., Ltd., Hainan Yishun Pharmaceutical Co., Ltd., Sichuan Feideli Pharmaceutical Co., Ltd., Guangxi Tiantianle Pharmaceutical Co., Ltd. and Quanzhou Luoshang, Fujian Province Six pharmaceutical companies in Shanshan Pharmaceutical Factory conducted follow-up inspections/flight inspections and found that the above-mentioned enterprises were suspected of having illegal and illegal production problems. They asked the local provincial bureaus to recover the relevant GMP certificates of the six involved enterprises, and investigated the alleged violations according to law.
Guangdong
60 batches of drugs were unqualified
On March 8, the Guangdong Provincial Food and Drug Administration issued the second report on the inspection and inspection of drugs in the second phase of 2018. The 60 batches of 50 varieties of drugs did not meet the requirements of the drug standards, and 57 batches were used in the use chain. batch.
A drug dealer GSP was withdrawn
On March 6, the Shenzhen Municipal Market and Quality Supervision and Management Committee issued the “Notice on Cancellation of GSP Certificatesâ€. Shenzhen Ruicaotang Pharmacy Co., Ltd. Shangshui Trail One Branch Drug Business Enterprise Seriously Violated the “Pharmaceutical Management Quality Management Regulations†It stipulates that it has been revoked its "Quality Management Certificate for Pharmaceutical Business Management" and assessed its credit rating as "serious breach of trust".
2 pharmaceutical companies illegally disclosed
Two pharmaceutical companies in Foshan City, Guangdong Province were publicly punished for producing/selling inferior drugs:
On March 6, the Foshan Municipal Food and Drug Administration issued the information disclosure form for the fourth case in 2018. The Chinese Medicine Pieces Factory of Guangdong Medicine Company was fined for producing the unqualified drug “Myrrhâ€. On March 9, Foshan Shunde District Market Supervision and Management Information Network released the first administrative punishment information disclosure form in March 2018. Foshan Mingren Pharmaceutical Co., Ltd. Longjiang Xixi Branch was suspected of selling inferior drugs "Lily" and " The Earth Dragon was ordered to correct the illegal act.
A pharmaceutical company was found to have 6 non-compliance facts
On March 6, the Guangdong Provincial Food and Drug Administration issued the first notice of the 20th year of 2018 on the rectification of the GSP for drugs, and ordered Guangdong Minshengtang Pharmaceutical Co., Ltd. to rectify within a time limit.
Guangxi
4 drug illegal advertisements were announced
On March 7, the Food and Drug Administration of Guangxi Zhuang Autonomous Region issued an announcement on illegal drugs, medical devices and health food advertisements, and announced the advertisements for illegal drugs, medical devices and health foods in December 2017.
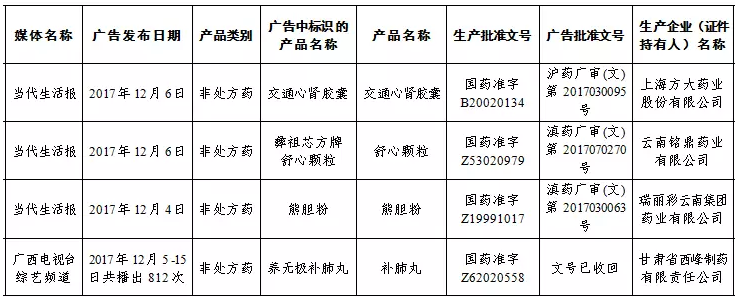
3 batches of drugs, 1 batch of Chinese medicine decoction pieces were found unqualified
On March 7, the Guangxi Zhuang Autonomous Region Food and Drug Administration issued a drug quality announcement (No. 2, 2018). Three batches of drug preparations and one batch of Chinese herbal medicine pieces did not meet the standard.
å½ç¥–芯方牌舒心粒 is suspended for sale
On March 6, the Food and Drug Administration of Guangxi Zhuang Autonomous Region issued an announcement that the “Zhuzuxin Fangxin Shuxin Granules†produced by Yunnan Mingding Pharmaceutical Co., Ltd. had serious exaggeration of product indications (functions and indications) in advertising. Content that deceives and misleads consumers is suspended for sale in Guangxi.
Anhui
10 kinds of medicines are not qualified!
On March 9th, Anhui Provincial Food and Drug Administration issued the 2nd issue of "Anhui Province Drug Quality Announcement" in 2018. 10 batches of 10 varieties of drugs did not meet the standard requirements, and one batch of samples was inspected and qualified, and did not meet the prescribed items. Including [traits], [check] moisture, weight difference, loading difference, dissolution, [leaching] and [content determination].
12 pharmaceutical manufacturers will be spot checked
On March 9, Anhui Provincial Food and Drug Administration issued the "2018 annual "double random and one open" targeted spot check work plan", 12 pharmaceutical production enterprises, 4 special drug management enterprises, and 20% of the drugs that undertake vaccine distribution business in 2018. Wholesale companies will be spot checked.
These pharmaceutical companies will be inspected!
On March 8, Anhui Tongling City Food and Drug Administration issued the "2018 Implementation Plan for the Flight Inspection of Pharmaceutical Enterprises in the City". The inspection targets are as follows:
The coverage rate of flight inspections for drug wholesale enterprises in the city is 100%, the coverage rate of flight inspections for drug retail chain enterprises in the city is 50%, and the coverage rate of flight inspections for drug retail enterprises in the city is not less than 10%. Puyang County and Yi'an District Bureaus shall determine the coverage of flight inspections for drug chain and retail enterprises within their respective jurisdictions. The inspection objects shall be randomly selected in principle, but one of the following circumstances shall be directly examined: (1) Daily management level Enterprises with low, chaotic purchase and sales channels; (2) enterprises that complain or report or have clues indicating that there are quality and safety risks; (3) enterprises that frequently change business licenses.
Guizhou
Suspected of serious violation of discipline, a deputy director of the Food and Drug Administration was investigated
On March 9, the Guizhou Provincial Commission for Discipline Inspection and Supervision announced that Luo Xiangbin, a member of the Party Committee and Deputy Director of the Guizhou Food and Drug Administration, was under review. Luo Xiangbin, a member of the Party Committee and Deputy Director of the Guizhou Food and Drug Administration, was suspected of serious violations of the law and is currently working on Receive disciplinary review and surveillance investigations.
Hainan
This pharmaceutical company received GMP
On March 7, Hainan Provincial Food and Drug Administration issued the No. 1 Announcement on the Recovering of Pharmaceutical GMP in 2018, and Hainan Yishun Pharmaceutical Co., Ltd. was reclaimed its “Pharmaceutical GMP Certificate†(Certificate No.: HI20170024).
Shanghai
4 drug dealers were required to rectify within a time limit
On March 7, Shanghai Qingpu District Market Supervision Administration issued the “Notice on Administrative Inspection Information of Pharmaceutical Retail Enterprises in the Region (February 2018)â€, and four enterprises were rectified within a time limit.
Hubei
There are 7 situations in pharmaceutical companies that will be seriously criticized.
On March 7, Hubei Xiaogan City Food and Drug Administration issued the “Methods for the Assessment and Application of Safety Credit Ratings of Pharmaceutical Enterprises in Xiaogan City (Trial)â€, which will be implemented from March 10, 2018, and will be valid for 2 years.
The seven conditions identified as severely untrustworthy are as follows:
1. The license has been revoked according to law in violation of the laws and regulations on drug supervision and management;
2. The administrative department of supervision was ordered to suspend the administrative punishment for serious violations of the law;
3. Being included in the joint disciplinary list by other departments;
4. Being punished by the supervision and management department for its illegal acts;
5. Forging alteration of administrative licenses, registration certificates, entry and exit quarantine certificates, approval documents, inspection reports, etc., when applying for administrative licenses, provide false certification materials or conceal facts to the food and drug supervision and administration department;
6. The person in charge of the enterprise and the person directly responsible shall be criminally dealt with due to drug safety violations;
7. Others have subjective intentions, selling counterfeit and inferior drugs, with serious plots and serious harm.
Jiangxi
2 pharmacies selling inferior drugs
On March 7, Jiangxi Xinyu City Market and Quality Supervision Administration issued the “Information Disclosure Form for Food and Drug Administration Punishment Cases in Jiangxi Province (February 2018)â€, Xinqing Pharmacy, Beihu East Road, Lishui District, Likang, Xinyu High-tech Zone The administrative punishment information of 2 pharmacies in the big pharmacy will be announced.
Shandong
2 pharmaceutical companies illegally disclosed
On March 6, Shandong Province Heze City Food and Drug Administration issued the "Administrative Punishment Information Disclosure (February)", Heze Zhongyuan Pharmacy Chain Co., Ltd. Jinhua Pharmacy purchased medicines from individuals who do not have the qualification for pharmaceutical business, and confiscated them according to law. The illegal income was 1374 yuan and the fine was 2748 yuan. The wholesale department of Heze Peony Medicine Co., Ltd. was not fined 12,000 yuan for not operating drugs according to the "Good Manufacturing Practices" and partially modifying and selling drugs to unqualified others.
Shanxi
2 pharmaceutical companies GMP received
On March 6, Shanxi Provincial Food and Drug Administration issued the "Repeated Drug GMP Certificate Announcement No. 33 of 2018", and recovered the "Drug GMP Certificate" from Shanxi Yunpeng Pharmaceutical Co., Ltd. and Shanxi Huawei Pharmaceutical Co., Ltd.
Shaanxi
18 batches of drug inspection failed
On March 6, Shaanxi Provincial Food and Drug Administration issued the "Shanxi Province Drug Quality Announcement", which announced the 18 batches of drugs that were found to be inconsistent with the regulations, and the items that did not meet the requirements were mainly for traits, identification, inspection, and content determination.
Beijing
Production of counterfeit drugs, a drug company was fined
On March 5th, Beijing Huairou District Food and Drug Administration issued the "Second Phase of Information Disclosure against Infringement of Counterfeit and Inferior Administrative Punishment Cases in 2018", and Beijing Yanbei Pieces Factory was fined for producing counterfeit drugs.
Jiangsu
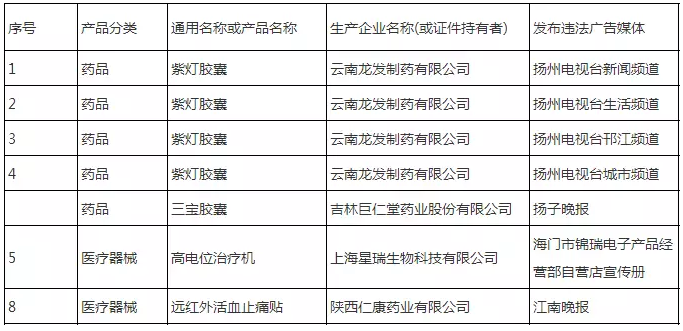
2 drugs, 2 devices illegal advertisements were announced
On March 5, Jiangsu Provincial Food and Drug Administration released the announcement of illegal drugs, medical devices and health food advertisements for December 2017, Ziguang Capsule of Yunnan Longfa Pharmaceutical Co., Ltd., and Sanbao Capsule of Jilin Jurentang Pharmaceutical Co., Ltd. The high-potential therapy machine of Shanghai Xingrui Biotechnology Co., Ltd. and the far-infrared blood-stasis and pain-relieving stickers of Shaanxi Renkang Pharmaceutical Co., Ltd. were announced.
Gansu
20 kinds of traditional Chinese medicine decoction pieces are supervised
On March 5, the Gansu Provincial Food and Drug Administration issued a notice to issue the "List of Key Regulators of Chinese Herbal Pieces in the First Quarter of 2018", including 20 varieties including Baiji, Mianma Guanzhong, Yam, and Seaweed.
Viral Transportation Medium Tube
Uses: used for the detection and sampling of influenza, hand, mouth, foot and other epidemic diseases
Inspection principle:
The combination of multiple antibiotics has broad-spectrum antibacterial and antifungal effects;
As a protein stabilizer, bovine serum albumin can increase the survival time and infection stability of the virus;
Buffers such as Hank's build a neutral environment, which helps to increase the survival time and infection stability of the virus;
Phenol red is an acid-base indicator, the discoloration area is 6.6 (yellow)~8.0 (red), and it is red at 7.2~7.4.
Steps:
1. According to the sampling requirements, use a sampling swab to collect samples.
2. Place the swab after collecting the sample into a sterile sampling tube.
3. Break the sampling swab rod that is higher than the sterile sampling tube.
4. Tighten the cap of the sterile sampling tube.
5. Label the sterile sampling tube with information as required.
For sample collection, transportation and storage.
Product advantages:
1. The virus discretion of the flocking swab is high to ensure the accuracy of the test results.
2. The samples are well sealed to ensure product transportation and safe storage.
3. Product instruction manual, product certificate
Product Details:
1. The product set includes a one-time Virus Sampling Tube (including preservation solution), a self-sealing bag, a sampling swab, and instructions.
2. Product specification: 100 sets/box, 8 boxes/box 3. Product weight: 0. 65kg/box, 13. 2kg/box
4. Packing size: 25. 5*23. 5*14. 5 boxes, 53*49*32/carton
Scope of application:
Work resumption testing, the best choice for large-scale population screening
Features:
1. Transport at room temperature, stably preserve viral RNA
2. Pre-packaged guanidine salt lysate can inactivate the new coronavirus, ensuring the safety of transportation and testing personnel 3. The large-capacity preservation solution can fully soak the swab head,
The sample size can be divided into three parts for testing and reserve samples respectively to meet the testing needs.
Scope of application:
Suspected cases, disease control testing, preferred by P3 laboratory
Viral Transportation Medium Tube,Sample Collection Tubes,Transport Nasal Swab With Tube,Virus Sampling Tube Nasopharyngeal Swab
Jilin Sinoscience Technology Co. LTD , https://www.jlgkscience.com