Vertex painkiller phase 2 clinically positive results
February 22, 2018 Source: WuXi PharmaTech
Window._bd_share_config={ "common":{ "bdSnsKey":{ },"bdText":"","bdMini":"2","bdMiniList":false,"bdPic":"","bdStyle":" 0","bdSize":"16"},"share":{ }};with(document)0[(getElementsByTagName('head')[0]||body).appendChild(createElement('script')) .src='http://bdimg.share.baidu.com/static/api/js/share.js?v=89860593.js?cdnversion='+~(-new Date()/36e5)];Vertex, a biopharmaceutical company, recently announced the positive results of a phase 2 clinical study of its sodium channel NaV1.8 inhibitor VX-150 in managing acute pain after hallux valgus correction. Compared with placebo, VX-150 treatment significantly ameliorated acute pain and was well tolerated.

This phase 2 study is the second proof-of-concept study of VX-150, and its positive results provide further validation for the treatment of pain with NaV1.8 inhibitors. The second phase 2 study of the VX-150 is currently underway, primarily for neuropathic pain, and is expected to be released in early 2019. Vertex recently launched a Phase 1 clinical study of the second NaV1.8 inhibitor VX-128 in healthy volunteers.
The phase 2 clinical study of the published data is a randomized, double-blind, placebo-controlled study that evaluated 243 patients with acute pain who used VX-150, hydrocodone + paracetamol Phenol or placebo in the treatment of acute pain after correcting hallux valgus. Eighty-two patients received placebo, 80 patients received VX-150, and 81 patients received hydrocodone + acetaminophen. Hydrocodone + acetaminophen (5 mg + 325 mg q6h) was used as a standard treatment reference group to aid in the evaluation of the potential therapeutic effect of VX-150, which was not statistically compared to the VX-150 group. The first oral dose of VX-150 was 1500 mg, followed by oral administration of 750 mg every 12 hours during the 48-hour treatment period.
The primary end point of the study was the time-weighted sum (SPID24) of the difference in pain intensity within 24 hours of treatment in patients treated with VX-150 as compared to placebo, as recorded by the Digital Pain Rating Scale (NPRS). An increase in the SPID24 value represents an improvement in pain relief. Secondary endpoints included safety and tolerability assessments as well as other efficacy measures, including SPID (SPID48) within 48 hours of treatment in the treatment group compared with the placebo group. Other pre-specified endpoint analyses included SPID24 and SPID48 for hydrocodone + acetaminophen compared to placebo.
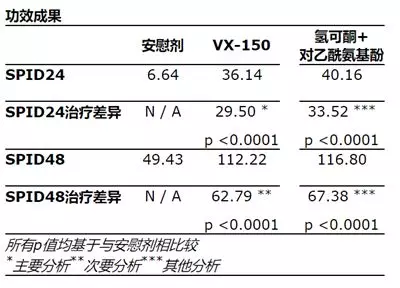
The results showed that the study reached its primary endpoint. There was a statistically significant improvement in SPID24 in patients treated with VX-150 compared to placebo. Patients treated with VX-150 and placebo had SPID24 values ​​of 36.14 and 6.64, respectively. Hydrocodone + acetaminophen has a SPID24 value of 40.16.

â–² Dr. Jeffrey Chodakewitz, Executive Vice President and Chief Medical Officer of Vertex (Source: Vertex Official Website)
Dr. Jeffrey Chodakewitz, Executive Vice President and Chief Medical Officer of Vertex, said: "The VX-150 and VX-128 represent an innovative approach to pain management. We are encouraged by the positive results of two VX-150 studies. Proving the potential of NaV1.8 inhibitors in the treatment of various pains. We look forward to generating more data from ongoing and planned two NaV1.8 inhibitor studies to inform future development plans for patients Bring these potentially new, transformative painkillers."
We are pleased to see the positive results of sodium channel NaV1.8 inhibitors in managing pain and expect new painkillers to alleviate patient suffering.
Reference materials:
[1] Vertex official website
[2] Vertex Announces Treatment with the NaV1.8 Inhibitor VX-150 Showed Significant Relief of Acute Pain in Phase II Study
Anesthesia Medical Co., Ltd. , https://www.medicaldiverse.com