Poco International Medical Trading (Shanghai) Co., Ltd. reported that a patient with a product in the subcutaneous implantable cardioverter defibrillator caused a memory failure and caused repeated output of atypical electrical energy, resulting in a patient implanted in the United States. On death, the manufacturer, Boston Scientific Corporation, voluntarily recalled the subcutaneous implantable cardioverter defibrillator (Registration No.: National Machinery Note 20153212410). The recall level is one level.
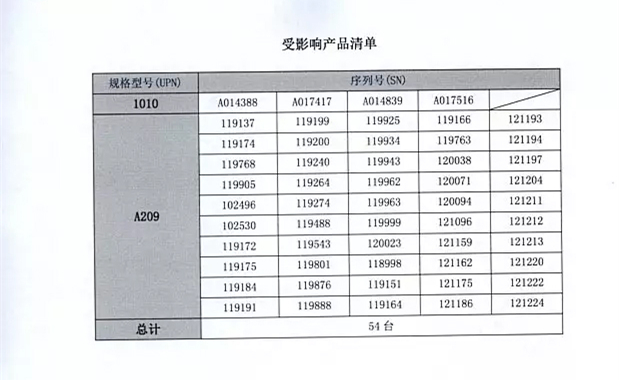
Detailed information on the model, specifications and batches of the products can be found in the Medical Device Recall Report Form.


China Extract Powder For Use As Dietary Supplement Extract Powder, Extract Powder Manufacturer
Shaanxi Kang New Pharmaceutical co., Ltd. , https://www.kangnewpharmas.com