Multi-stage mass spectrometry for in-depth analysis of compound structures
Drug Impurity Analysis Solution--Ion Trap Multi-Level Liquid Quality
Preface
Drug impurities have become an important concern of drug regulatory agencies at home and abroad because of their potential impact on drug quality, safety and efficacy. With the expansion of the export scale of China's pharmaceutical products, understanding the control requirements of drug impurities in foreign regulatory markets and strengthening the analysis and control of drug impurities have become a common concern of domestic pharmaceutical manufacturers.
Any substance that affects the purity of a drug is collectively referred to as an impurity. The technical requirements for human drug registration require the International Harmonization Council (ICH) to define an impurity as any component of a drug that is inconsistent with the drug. Impurities in the drug will reduce the efficacy, affect the stability of the drug, and some may even be harmful to human health or produce other toxic side effects. Therefore, the detection of related substances, control of purity to ensure the safety and effectiveness of medication, is very important to ensure the quality of the drug.
Impurity spectrum analysis refers to the distribution of known and unknown impurities in the study drug, and analyzes the source and destination of impurities in the drug. Through the study of the impurity spectrum, the safety of the drug can be comprehensively evaluated. For the drug production stage, the impurity spectrum study can establish a complete and reliable impurity analysis method in the process, monitor the change of impurities in the key steps of the process, verify the impurity analysis method and transfer to QA/QC, for the drug development stage, it needs to The impurities in the art development process are identified and characterized and the source of the impurities is further confirmed. Based on the analysis results, the R&D personnel can evaluate the safety of the drug and the consistency with the original drug, and further optimize the process according to the source of impurities to reduce or eliminate the generation of impurities. .
The organic impurities mainly originate from the starting materials and the impurities contained in the raw materials, the synthetic intermediates and side reaction products brought in the production process, the degradation products produced by the finished products during the storage process, and the impurities or auxiliary materials brought in by the auxiliary materials are degraded by interaction. Impurities, and impurities produced by the interaction of the drug and the package. The content of organic impurities in the finished drug is usually very low, and the preparation and purification require a lot of manpower and material resources. The conventional analytical method is difficult to cope with the multi-component, low-content impurity structure analysis work. Ion trap mass spectrometer, because of its sensitive MSn ability, high-throughput routine analysis of complex samples, high-quality structural information, fast positive and negative ion switching technology, fast positive and negative ion switching technology, stable, reliable, easy to operate, widely used In the field of drug impurity research, clinical and forensic identification, natural compound identification, metabolite identification and other fields.
Impurity analysis workflow
The impurities in the drug are often very small. In the presence of a large amount of the parent drug, the detection of trace impurities is a major challenge to mass spectrometry. After collecting the mass spectrometry information of the impurities, the structure of these process impurities or degradation products is effective. The analysis is another big challenge. Thermo's ion trap technology effectively solves the above impurity research problems. Its high sensitivity and wide dynamic range enable the collection of effective mass spectral information of trace impurities. The ion trap's multi-stage mass spectrometry capability and powerful analytical software can deeply penetrate the impurity structure. Analytical, combined with the efficient separation of chromatography and professional structural analysis software, Thermo has established a solution for impurity analysis.
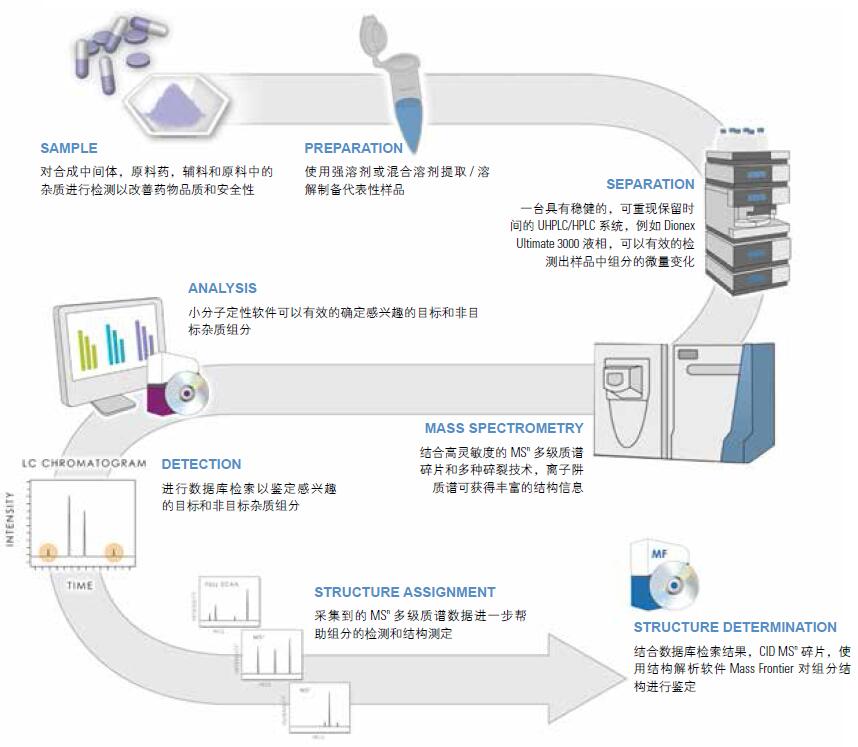
Figure 1. Ion trap-based impurity analysis process
Introduction to ion trap mass spectrometry
Ion trap mass spectrometry
Ion traps are time-series mass spectrometry techniques. The capture, isolation, excitation, and ejection of compound ions are all done in the well. Because of this, the ion trap repeats this scanning process to complete multi-stage fragmentation (MS n ).
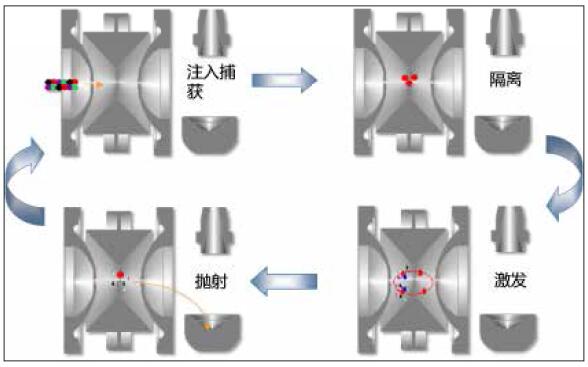
Figure 2. Ion trap time series scan
The stable region in the ion trap can be calculated by the Marshall equation. The compound ions move at a specific frequency under a specific RF voltage, increase the RF voltage, and the frequency (q value) of the ions will increase, gradually increasing the RF, and making the ions low. From m/z to high m/z, gradually go away and complete the scan.

Figure 3. Ion trap scanning principle
Thermo patented linear ion trap mass spectrometry
As the leader in ion trap mass spectrometry, Thermo's patented linear ion trap provides the fastest speed while maintaining excellent sensitivity.
â— The ions gather along the centerline, which also reduces the space charge effect and greatly increases the ion capacity;
â— Dual radial detector design detects ions at the same time, the ion detection speed is faster, the response intensity is higher, and the signal is more stable;
â— The ion trapping efficiency is higher, the sensitivity is higher, and more series of fragmentation are performed;
Proteomics
â— Maximum sequence coverage from excellent cycle times and sensitivity
â— Automatic MSn trigger data dependent neutral loss scan
â— Flexible ionization mode, inlet and chromatography solutions
Quantification of drug development
â— Excellent ion statistics make the quantification stable and reliable
â— Collision energy normalization for maximum work efficiency
â— Provide simultaneous qualitative and quantitative data for regulated laboratories
Forensic and clinical analysis
â— Durable stability for reliable quantification of complex matrix samples
â— Simultaneous quantification and characterization in positive and negative ion modes
â— Easy operation to improve laboratory efficiency
Metabolite identification
â— Excellent MSn spectral quality and full scan acquisition technology
â— Low concentration metabolite structure confirmation, no need to understand MRM information
â— Data dependency and dynamic exclusion techniques get the most information from complex samples
data collection
Multiple scanning methods
The Thermo Ion Trap offers a rich sampling mode that combines common templates to help quickly build analytical methods and expands to meet the needs of in-depth analysis.
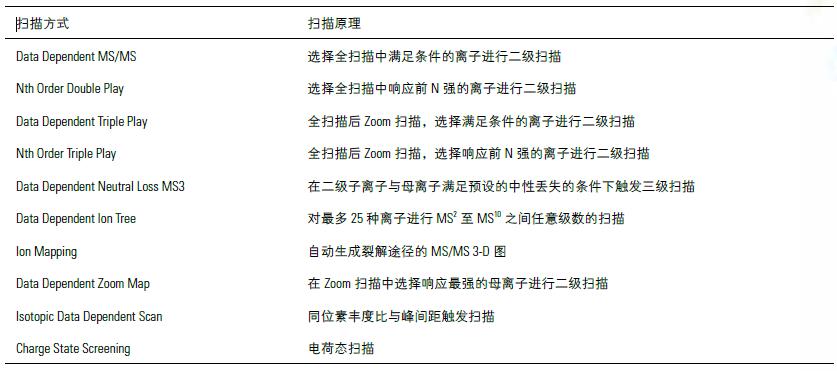
Note: Zoom scanning is an ion trap scanning method that increases the resolution by reducing the scanning speed.
Target impurity analysis
In the impurity research process, it is usually necessary to analyze the known or target impurities in the drug. The ion trap selective reaction monitoring (SRM) can scan the impurity-specific parent ions and daughter ions to ensure the specificity and accuracy of the results, and the continuous reaction. Monitoring (CRM) can scan the impurity-specific parent ions, daughter ions and multi-level ions for higher sensitivity and anti-interference ability. In the figure below, SRM is used to scan the parent ion m/z 258 and product ions of the target compound. It can be seen that there is a clear peak at 2.65 min. After further scanning the tertiary compound m/z 133 of the target compound by CRM, The background interference of 2.65min compound was significantly reduced, and the signal-to-noise ratio was greatly improved.
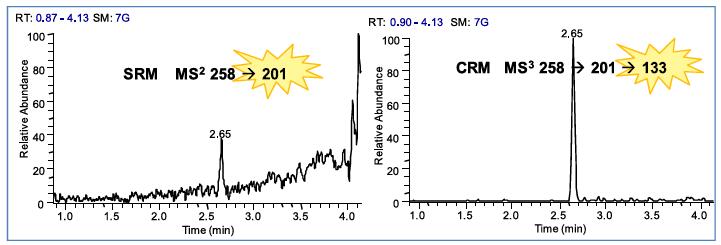
Figure 4. Multistage mass spectrometry provides greater specificity and sensitivity
Unknown impurity analysis
The identification of unknown impurities is a very important part of the impurity research work. Because the structure is unknown, how to collect accurate impurity precursor ions and multi-stage mass spectrometry information becomes the premise and key of structural analysis. Data-associated multi-level mass spectrometry and dynamic scanning capabilities enable the acquisition of unknown impurity signals.
As shown in the figure below, the ion trap obtains the mass spectrometry information of the compound through one-stage scanning, and automatically selects the parent ion for multi-stage mass spectrometry scanning according to the intensity of the mass spectrometry response. The impurity multi-stage mass spectrometry information can be obtained by one injection.
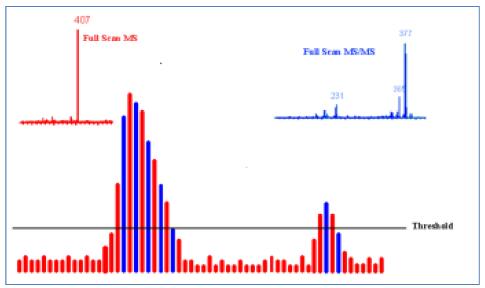
Figure 5. Data correlation scan simultaneously collecting known and unknown impurities
In the analysis of loperamide drug by data correlation scanning, the ion trap obtains the first-order mass spectrometry signal through the full sweep, and then the second-stage fragmentation of the most responsive loperamide precursor ion, and then automatically selects m/ The two main secondary ions of z 266 and m/z 210 undergo three-stage fragmentation, followed by a four-stage fragmentation of the tertiary ions m/z 210 and m/z 238 produced by the secondary ion m/z 266. The data correlation scan can automatically complete the multi-stage mass spectrometry process in one injection, without knowing the compound information in the sample in advance, and improving the analysis throughput and data information.
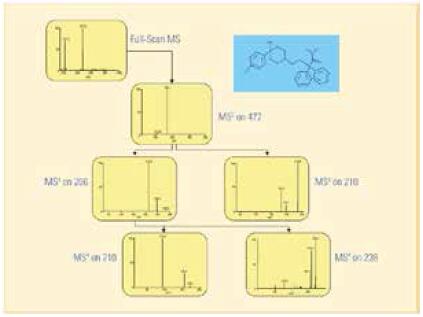
Figure 6. Data correlation scan for loperamide multistage mass spectrometry data
Dynamic exclusion function: In the study of drug impurities, the transfer process from the liquid phase method to the liquid method is often involved, and the liquid-based method is often unable to separate all impurities in a short time because the mobile phase is different from the liquid phase. With dynamic exclusion function, it can multi-stage fragmentation of all co-eluting impurities after opening, in order to cope with the fact that the chromatogram does not completely separate the impurities.
In the figure below, the compound m/z 452 and m/z 407 co-eluted. After the dynamic elimination was started, the instrument firstly split the m/z 407 with high response, then put m/z 407 into the exclusion. In the list, in the next full sweep, the higher response m/z 407 is used as the exclusion, and instead the weak response m/z 452 is selected for secondary fragmentation. Therefore, in a chromatographic peak, even if multiple compounds are present, dynamic exclusion ensures multi-stage mass spectrometry signals for each compound.
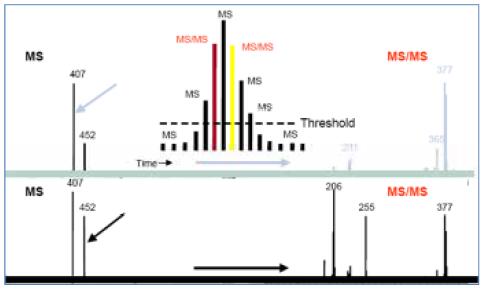
Figure 7. Dynamic exclusion of collected co-eluting impurities
data analysis
There are many kinds of impurities. Even compounds with the same element composition may have completely different structures. Therefore, it is necessary to effectively analyze the collected data and judge the estimated structural formula. Mass FrontierTM is the management, evaluation and analysis of the mass spectrum. Provides sophisticated features. It can be widely used in the identification of small molecule structures, and its application range covers a wide range of fields including drug development, impurity analysis and identification of metabolites in toxicological research.
Figure 8. Mass Frontier structure analysis software
MassFrontier professional structure analysis software
With Massfrontier, researchers can easily get the following information from the collected data:
â— Cracking mechanism of the compound
Because the structure of the impurity and the structure of the parent drug are often similar or have a relationship, understanding the cleavage law of the parent drug can help determine the fragment structure of the impurity and further infer the structure of the impurity.
Previous methods relied solely on the individual experience of the researcher, or purely theoretical calculations, which limited the reproducibility and accuracy of data analysis.
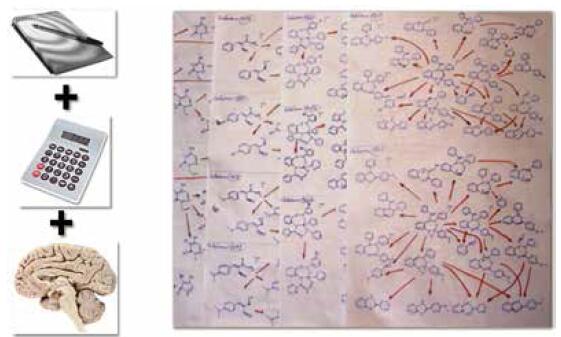
Figure 9. Traditional structure analysis method
The Fragmentation LibraryTM Fragmentation Mechanism Library contained in Mass Frontier is collected from all existing publications on mass spectrometry and covers almost all published literature. Each mechanism, along with its chemical structure, is manually and automatically checked and then stored in the library together with additional information such as title, author, and information source. The information collected by the spectral library, combined with general ionization, fragmentation and recombination rules, provides a strong guarantee for the prediction of the fragmentation mechanism. Mass Frontier also allows users to create spectral libraries based on customer-defined fragmentation mechanisms and to learn and apply these mechanisms to intelligently predict fragmented molecules.
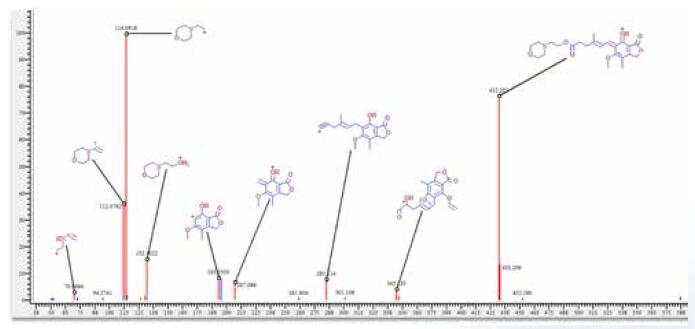
Figure 10. Fragmentation Library provides mycophenolate ester cracking mechanism based on published literature
â— Seizure of related substances
Some impurity components may be neglected by researchers because the signal response is too low, or the chromatographic peak formed is not sharp enough, but the low content of toxic impurities seriously affects drug safety. Leak detection will not only greatly affect the safety of medication. It will also affect the new drug declaration indirectly.
MassFrontier's Fragment Ion Search (FISh) allows for the rapid screening of structurally similar compounds by filtering a set of cleavage fragments, either by theoretical lysis prediction or by experimental MSn mass spectral tree, by searching for impurities and parental fragments. Accurately identify all relevant impurities.
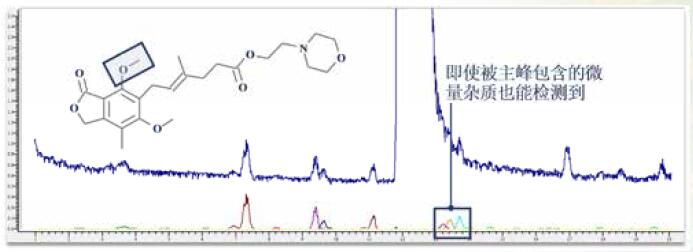
Figure 11. Fragment Ion Search (FISh) retrieves traces of mycophenolate mofetil
Application of ion trap mass spectrometry in the analysis of drug impurities
Comprehensive identification of adefovir dipivoxil impurity spectrum by linear ion trap multiple dissociation techniques
Analysis of trace impurity components in pharmaceutical intermediates using LCQ ion trap mass spectrometer and Mass Frontier software
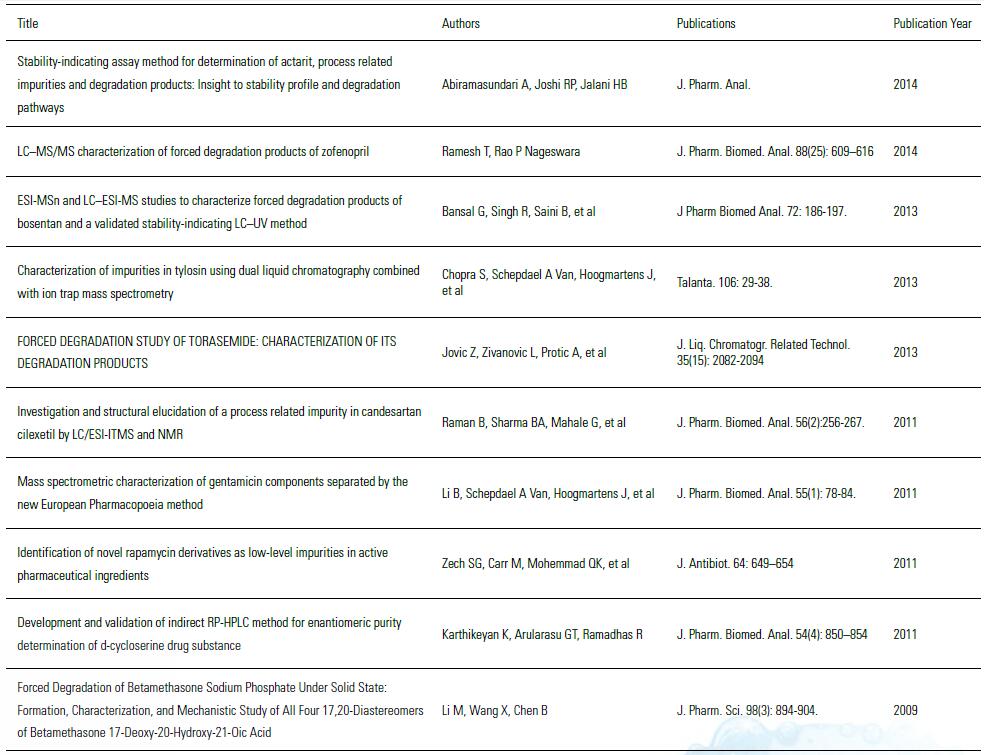
Drug impurity analysis part customer article list
appendix
Classification of Impurities: The ICH guidelines classify impurities into three categories: organic impurities, inorganic impurities, and residual solvents.
â— Organic impurities:
Including the impurities and degradation products introduced in the process, they mainly originate from the starting materials and impurities contained in the raw materials, the synthetic intermediates and side reaction products brought in the production process, and the degradation products produced during the storage process of the finished products. Impurities generated by the degradation of impurities or excipients introduced in the process, and impurities generated by the interaction of the drug and the package material. The chemical structure of organic impurities is similar to or related to the molecular formula of the active ingredient and is often referred to as the related substance.
The determination of the limit of organic impurities: the original research drug has generally done a comprehensive study on the safety of impurities, so for generic drugs, it is necessary to carry out sufficient impurity comparison and research with the original drug, analyze the type and content of impurities, and combine the original drug The quality standard, the control of impurities, if the impurities of the generic drug are different from the original drug due to the difference in process and prescription, the new impurities in the generic drug need to be within the limits of Tables 1-1 and 1-2, if new impurities Exceeding the limits in the table, or the impurity content of the generic drug is significantly higher than the original product, it is necessary to optimize the prescription and process to reduce the impurity content to within the specified quality control limit. If it cannot be lowered to the specified limit, it is necessary to exceed the limit. The impurities were investigated for safety.
Table 2. Impurity limits for APIs

Table 3. Impurity limits for formulations
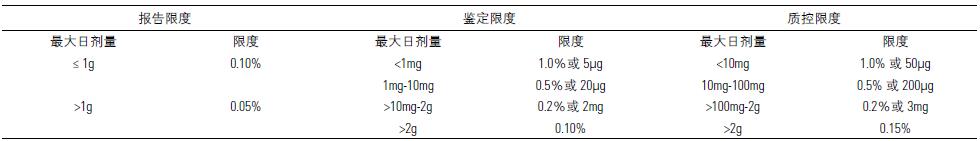
One of the organic impurities that directly destroys DNA activity and increases the risk of cancer in patients with genotoxic drugs is collectively referred to as genotoxic impurities, including impurities produced in the production process or degradation products produced during storage. According to FDA and EMA regulations, genes The quality control limits for toxic impurities are much lower than for general related substances. Recommended acceptance thresholds are in the FDA and EMA guidelines.
â— Inorganic impurities:
Impurities generated during the production or delivery of APIs and preparations are generally known. Sources may include reagents in the production process, such as catalysts, ligands, etc.; metals introduced at other stages, such as reaction vessels, transfer piping, filter materials, etc.
â— Residual solvent:
The organic solvent used in the production process of the drug substance and the preparation, for example, the final purification and crystallization process of the drug substance may leave a small amount of organic solvent, which is difficult to remove completely. Such residual solvent is often harmful to humans or the environment, or causes degradation of the drug substance. Residual solvents generally have known toxicity, and ICH, USP and EP all specify a limit for residual solvents. The ICH guidelines classify residual solvents in drugs into four categories. One type of solvent (benzene, carbon tetrachloride, etc.) is the most toxic and should be avoided. The second type of solvent (acetonitrile, chloroform, cyclohexane, etc.) is more toxic. It should be restricted. The three types of solvents (acetic acid, acetone, formic acid, etc.) should be restricted according to the GMP or other quality requirements. The four types of solvents (petroleum ether, trifluoroacetic acid, etc.) do not have sufficient toxicological information.
â— Other:
Exogenous pollution: microbes, endotoxin, etc.
Polymorph: Crystalline impurities
Corresponding isomer: chiral impurity
Editor: Zhou Zhe, Jiang Yan
Proofreading: Jiang Yan
Thermo Mass Spectrometry Resource Center
â— Under the Thermo Scientific brand of Chromatography and Mass Spectrometry, the predecessor of the famous mass spectrometer - the Finnigan mass spectrometer manufacturer. For more than 40 years since its founding in 1967, it has maintained its world leading position in mass spectrometry with unparalleled research. It has become the most reliable partner for the vast majority of analytical and research workers in the fields of agriculture, environmental protection, medical and health, inspection and quarantine, petrochemical, pharmaceutical, public security and life sciences.
â— More information on Thermo ion trap mass spectrometry is available
Http://
XuChang Youjia Food Product Development Co., LTD , https://www.yjgrain.com