Crystal structure of glucagon receptor reveals B-type GPCR signal transduction mechanism
January 04, 2018 Source: Shanghai Institute of Materia Medica
Window._bd_share_config={ "common":{ "bdSnsKey":{ },"bdText":"","bdMini":"2","bdMiniList":false,"bdPic":"","bdStyle":" 0","bdSize":"16"},"share":{ }};with(document)0[(getElementsByTagName('head')[0]||body).appendChild(createElement('script')) .src='http://bdimg.share.baidu.com/static/api/js/share.js?v=89860593.js?cdnversion='+~(-new Date()/36e5)];- Analysis of the three-dimensional structure of the glucagon receptor and polypeptide ligand complexes
Another important development in the field of GPCR research
Recently, the Shanghai Institute of Materia Medica, Chinese Academy of Sciences, has made another important advance in the study of the structure and function of G protein-coupled receptor (GPCR): the first measurement of glucagon receptor (Glucagon receptor) , GCGR) The three-dimensional structure of the full-length protein-polypeptide ligand complex reveals the specific recognition and activation regulation mechanism of the receptor on cell signaling molecules. This achievement will help to understand the structural biology of the physiological effects of type B GPCRs and accelerate the development of new drugs for the treatment of type 2 diabetes. Relevant research papers were published on the international top academic journal Nature on January 4, 2018. The authors were researcher Wu Lili and researcher Zhao Qiang.
GPCR is the largest family of membrane receptor proteins in human body and plays an important role in cell signal transduction. GPCR is closely related to human diseases, and more than 40% of listed drugs currently target GPCR. According to their similarities, GPCRs can be divided into four types: A, B, C, and F. Type B GPCRs include a variety of important receptor proteins, such as GCGR, which recognize and bind peptide hormones and are essential for maintaining hormone balance in the body. Such receptors comprise an extracellular domain and a transmembrane domain, both of which are involved in the recognition of cellular signals. Since it is extremely difficult to obtain a stable and intact type B GPCR protein (especially a complex of a type B GPCR and a polypeptide ligand), its structural study is extremely challenging.
GCGR is involved in the regulation of blood glucose homeostasis and is an important target for the treatment of type 2 diabetes drugs. The lack of structural information not only severely restricts the understanding of the receptor signal recognition and transduction mechanism, but also greatly affects the targeting of GCGR. Drug development? There are currently no listed drugs. In 2017, three research groups led by Wu Yili, Wang Mingwei and Jiang Hualiang of the Shanghai Institute of Materia Medica of the Chinese Academy of Sciences collaborated to analyze the full-length GCGR protein simultaneously with a small molecule allosteric modulator (NNC0640) and antagonist antibody (mAb1) antigen. Combining the crystal structure of the fragment-bound complex, the three-dimensional structure of the full-length B-type GPCR protein was presented for the first time at a higher resolution level, and the cooperative regulation mechanism of the activation of different domains of the receptor was revealed. A key step in the B-type GPCR signal transduction mechanism.
Later, the relevant research team of the Shanghai Institute of Materia Medica of the Chinese Academy of Sciences again collaborated to solve the crystal structure of the complex of the full-length GCGR combined with the glucagon analog NNC1702, revealing the fine combination of the B-type GPCR and the peptide ligand. mode. Researcher Wu Lili, the project leader, said: "This achievement is another important development in our research on the structure and function of B-type GPCR. The clarification of the interaction mode between GCGR and peptide ligands not only helps to understand the B-type GPCR on cells. The identification mechanism of signaling molecules and the most accurate structural templates to date for GCGR-targeted drug design will greatly facilitate the development of new drugs for the treatment of type 2 diabetes."
In a previous study, the team members found that peptides linked to the extracellular domain and transmembrane domain of GCGR play a key role in the regulation of receptor activation through interaction with other regions of the receptor protein. The structure of the complex of GCGR binding to the polypeptide ligand NNC1702 was analyzed and compared with the previously analyzed full-length GCGR structure. They further found that the linker peptide undergoes a significant conformational change in the receptor-binding polypeptide ligand, and its secondary The structure is transformed from a beta sheet to an alpha helix, and with the migration of the structure, the relative orientation between the two domains of the receptor changes greatly, thereby promoting the tight binding of the receptor to the polypeptide ligand, resulting in receptor activation. In addition, the linker peptide finely regulates the conformation of the receptor transmembrane domain by interaction with the mid-region of the polypeptide ligand, thereby regulating receptor activation. Researcher Zhao Qiang, co-author of the paper, said: "This discovery is truly amazing. Although it contains only 12 amino acids, this linker peptide plays such an important role. This has never been found in past GPCR structural studies. However, we have a deeper understanding of the signal regulation mechanism of B-type GPCR."
Based on the complex structure of GCGR combined with NNC1702, the team also carried out a series of functional studies using receptor-ligand competition binding, computer simulation and two-electron resonance to clarify the conformation of GCGR under different functional states. The dynamic changes and the in-depth exploration of the regulation mechanism of receptor activation. The research was supported by several research groups including Shanghai Pharmaceutical Research Institute, Fudan University and Shanghai University of Science and Technology. Academician Jiang Hualiang, one of the main partners of the project and director of the Shanghai Institute of Materia Medica, stressed: "This is not only another major research achievement of the Shanghai Institute of Drugs GPCR research team, but also a GPCR research highland has been in Shanghai Science and Technology Innovation Center. The core area of ​​construction - the rise of Zhangjiang Hi-Tech Park.
The first author of the research paper is graduate student Zhang Haonan. The main partners of the project are Wang Mingwei, researcher of Shanghai Institute of Materia Medica, Chinese Academy of Sciences, researcher Yang Dehua, Professor Raymond Stevens of iHuman Institute of Shanghai University of Science and Technology, Steffen Reedtz-Runge of Novo Nordisk, Denmark Dr. Oliver Ernst from the University of Toronto, Canada, Dr. Michael Hanson from the American GPCR Research Alliance, Dr. Yang Linlin from Zhengzhou University, and Professor Yang Huaiyu from East China Normal University. The research was funded by the Chinese Academy of Sciences, the National Natural Science Foundation of China, the Shanghai Science and Technology Development Fund, and the Shanghai Municipal Education Commission.
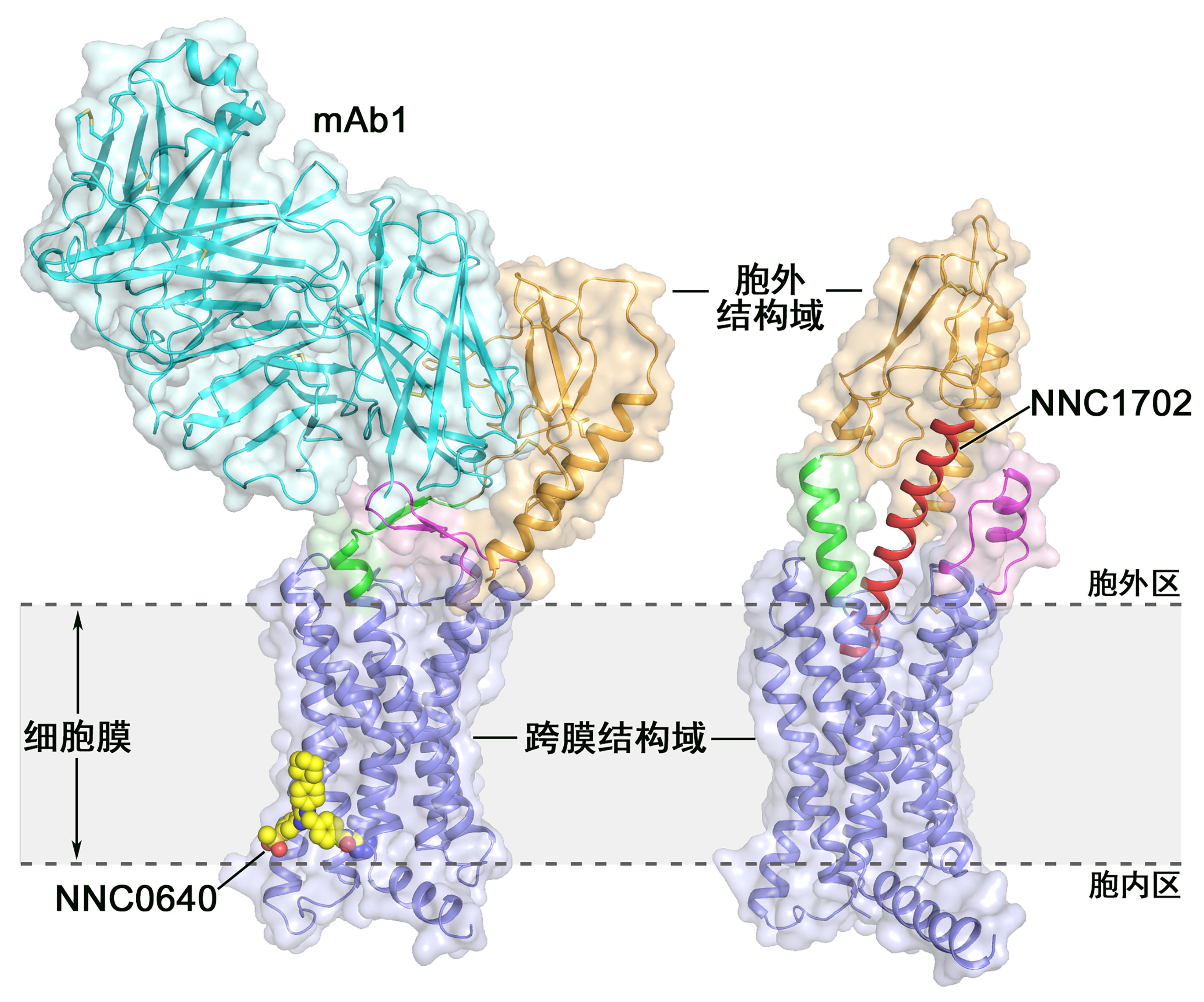
Schematic diagram of full-length GCGR structure: GCGR is involved in the regulation of blood glucose homeostasis and is an important target for the treatment of type 2 diabetes drugs.
The left panel shows the crystal structure of the complex of the full-length GCGR protein combined with the small molecule allosteric modulator NNC0640 and the antagonist antibody mAb1;
The right panel shows the crystal structure of the complex of the full-length GCGR protein bound to the polypeptide ligand NNC1702.
The two structures are represented by a streamer map and a surface map. The transmembrane domain of GCGR is blue, the extracellular domain is orange, the connecting peptide is green, the first extracellular loop is magenta, and NNC1702 is red (right) ), NNC0640 is yellow (left) and antibody mAb1 is blue-green (left). The cell membrane is indicated by a gray area.
Article link: https://
Blackhead Suction Tool,Best Blackhead Remover,Electric Blackhead Remover,Best Blackhead Suction Remover
Shenzhen Jie Zhong Lian Investment Co., Ltd. , https://www.szmeizons.com