Treatment of rare tumors, FDA approved the first peptide receptor radionuclide therapy
January 29, 2018 Source: WuXi PharmaTech
Window._bd_share_config={ "common":{ "bdSnsKey":{ },"bdText":"","bdMini":"2","bdMiniList":false,"bdPic":"","bdStyle":" 0","bdSize":"16"},"share":{ }};with(document)0[(getElementsByTagName('head')[0]||body).appendChild(createElement('script')) .src='http://bdimg.share.baidu.com/static/api/js/share.js?v=89860593.js?cdnversion='+~(-new Date()/36e5)];Recently, the US FDA announced that it has approved Lutathera (lutetium Lu 177 dotatate) of Novartis Group Advanced Accelerator Applications for the treatment of somatostatin receptor-positive gastrointestinal pancreatic neuroendocrine tumors (GEP-NETs). This is also the first polypeptide receptor radionuclide therapy (PRRT) approved by the US FDA.

Neuroendocrine tumors (NETs) are a rare type of tumor that can cause neuroendocrine cells in many organs such as the gastrointestinal tract, pancreas, and lungs. Patients with this type of tumor have different symptoms. Some patients develop symptoms due to excessive hormone production in the body, while others can be asymptomatic for many years. Although these types of tumors are not common, once they become ill, they will have a great impact on the patient's life. It is estimated that once the tumor differentiates and distant metastasis occurs, the 5-year survival rate of these patients will be only 35. %. Innovative therapy has become a lifeline for these patients.
The approved Lutathera is a radiopharmaceutical that binds to the somatostatin receptor on the surface of tumor cells and enters cells to cause damage to tumor cells through radioactivity. Previously, it was granted orphan drug status by the US FDA and was also eligible for priority review.
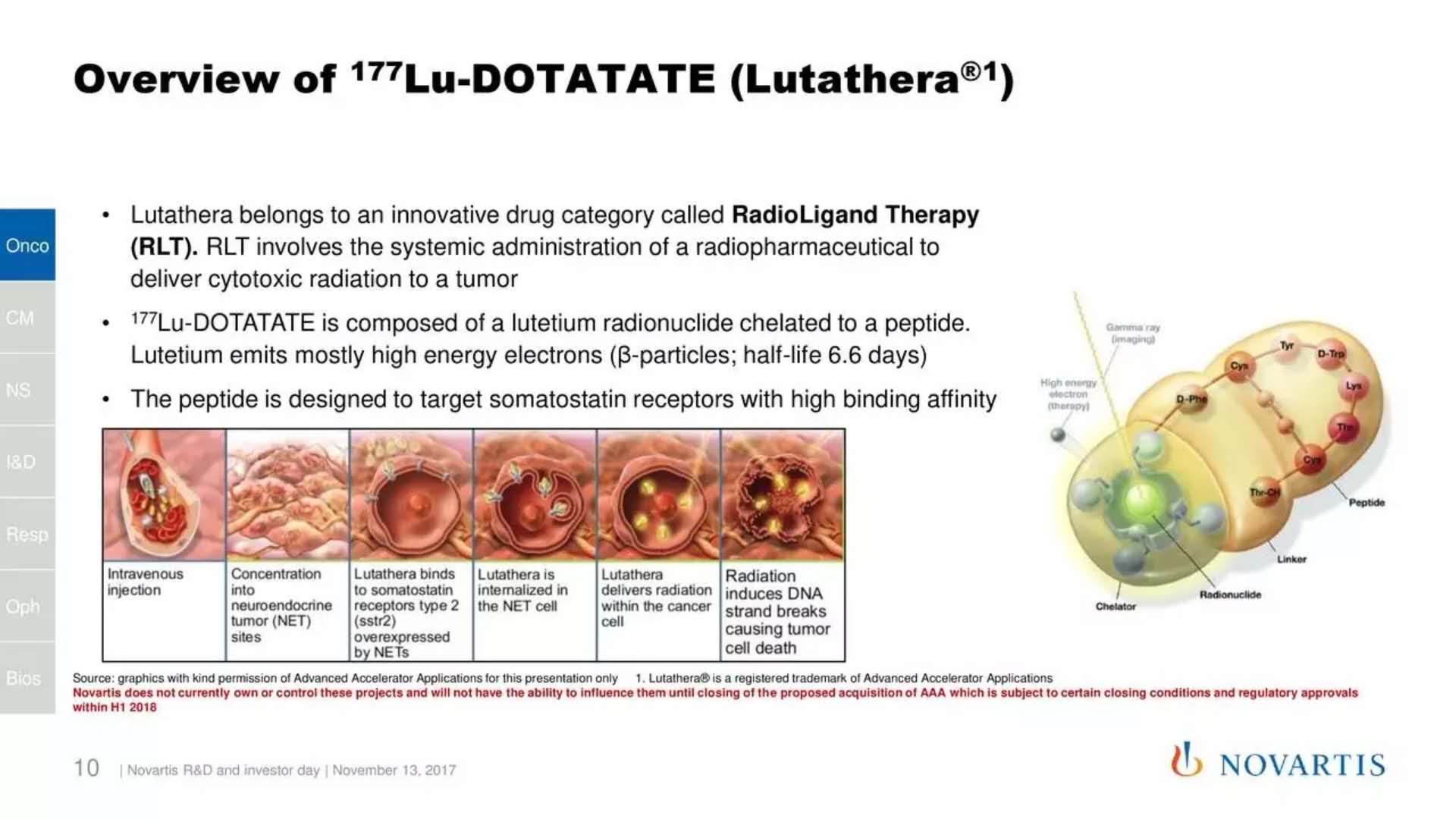
â–² Lutathera's mechanism of action (Source: Novartis)
The efficacy of this new drug has been validated in two key phase 3 clinical trials. In the first trial, in combination with the best standard of care (30 mg octreotide LA per week), Lutathera showed good results, reducing the risk of disease progression or death by 79% (HR 0.21, 95% CI: 0.13-0.32; p<0.001); in the second trial, Lutathera made 16% of patients in a subgroup of 360 patients with GEP-NETs in 1214 patients with somatostatin receptor-positive tumors. Some tumors shrink. It is worth mentioning that the patients enrolled in the second clinical trial are part of the FDA's "expanded access" program, which allows patients with severe and life-threatening illnesses to receive new drugs for treatment. Based on data from these two trials, the FDA also decided to approve the new drug to market for the benefit of patients.
“GEP-NETs are a rare type of cancer that will continue to grow after initial treatment failure, and patients have limited treatment options,†said Richard, Director of the Center for Excellence in Oncology and Executive Director of the Office of Hematology and Oncology Products. Dr. Pazdur said: "This approval has brought new treatments to patients with these rare cancers. This also demonstrates that the FDA is willing to consider the data in the 'expanding use' program to support the approval of new therapies."
â–² Lutathera is expected to bring more treatment to patients (Source: Carcinoid Cancer Foundation)
“An important achievement of Lutathera's approved logo is also an important innovation for the NET cancer community,†said Dr. Susanne Schaffert, Chairman and President of Advanced Accelerator Applications: “For more than 30 years, Novartis has been developing NET and carcinoid syndrome. Therapy supports the NET community. We will work together to advance innovative nuclear medicine therapies to treat a variety of tumor types including NET. I don't think there is any better way to celebrate the cooperation and future of our two companies."
We congratulate Novartis and Advanced Accelerator Applications for winning the approval of this new drug, and we look forward to more innovative treatments that will continue to benefit patients.
Reference materials:
[1] Advanced Accelerator Applications Receives FDA Approval for Lutathera® for Treatment of Gastroenteropancreatic Neuroendocrine Tumors
[2] FDA approves new treatment for certain digestive tract cancers
China Extract Powder For Use As Dietary Supplement Extract Powder, Extract Powder Manufacturer
Shaanxi Kang New Pharmaceutical co., Ltd. , https://www.anabolicsteriod.com