Abelvieus uterine fibroids new drug phase 3 clinical trial reached the primary end point
March 14, 2018 Source: WuXi PharmaTech
Window._bd_share_config={ "common":{ "bdSnsKey":{ },"bdText":"","bdMini":"2","bdMiniList":false,"bdPic":"","bdStyle":" 0","bdSize":"16"},"share":{ }};with(document)0[(getElementsByTagName('head')[0]||body).appendChild(createElement('script')) .src='http://bdimg.share.baidu.com/static/api/js/share.js?v=89860593.js?cdnversion='+~(-new Date()/36e5)];AbbVie and Neurocrine Biosciences today announced that the uterine fibroids reached the primary end point in the phase III clinical study ELARIS UF-II (M12-817). This study is a repetitive test of another key phase 3 trial, ELARIS UF-I (M12-815), whose results were published in February 2018. The study confirmed the results of the first trial, significantly reducing severe menstrual bleeding in patients with uterine fibroids, reaching the primary end point of the study.

Uterine fibroids, also known as leiomyomas or fibroids, are non-cancerous, hormone-responsive uterine muscle tissue tumors. Uterine fibroids are the most common abnormal growth in the female pelvis, affecting approximately 20-80% of women under the age of 50. The size, shape, number and location of fibroids vary. Patients with uterine fibroids may be asymptomatic, but in some women, fibroids can cause serious problems such as severe menstrual bleeding, dysmenorrhea, and even pregnancy difficulties.
Elagolix is ​​a gonadotropin-releasing hormone (GnRH) receptor antagonist, an oral short-acting molecule that blocks endogenous GnRH signaling by competing with the GnRH receptor in the pituitary gland. Rapid reversible and dose-dependent inhibition of luteinizing hormone (LH) and follicle stimulating hormone (FSH) secretion during treatment can result in reduced production of ovarian sex hormones, estradiol and progesterone. Elagolix is ​​currently being studied in patients with ovarian sex hormone-mediated diseases such as uterine fibroids and endometriosis. To date, Elagolix has conducted studies in more than 40 clinical trials involving a total of more than 3,700 subjects.
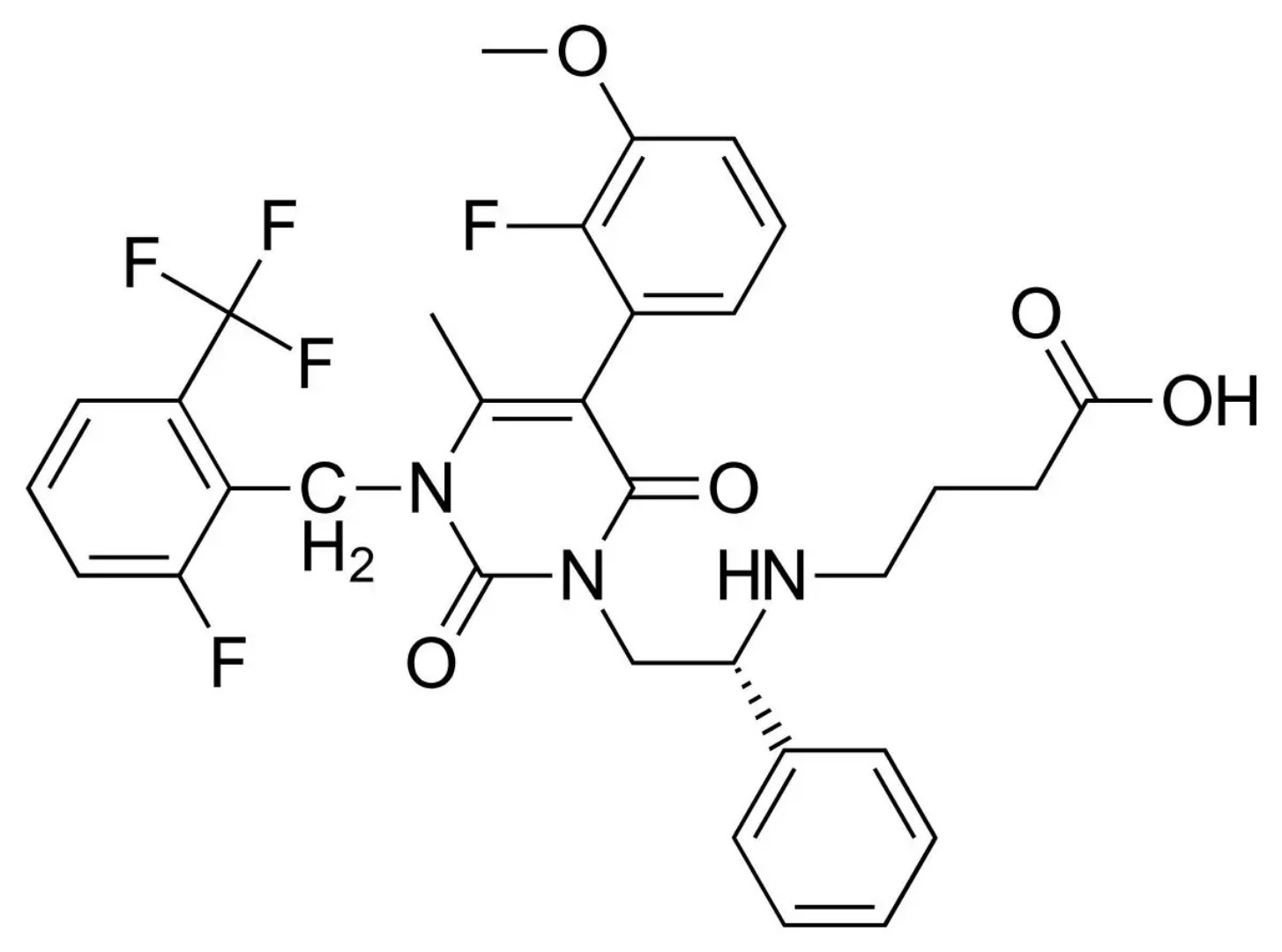
â–²Elagolix's molecular structure (Source: By Vaccinationist (PubChem) [Public domain or Public domain], via Wikimedia Commons)
ELARIS UF-II is the second of two 6-month repeat phase 3 clinical trials evaluating the safety and tolerability of elagolix and elagolix combined with low-dose hormonal therapy (estradiol/norethrel acetate) And efficacy. The study subjects were premenopausal women diagnosed with uterine fibroids by transvaginal and transabdominal pelvic ultrasonography (TVU) between the ages of 18 and 51. The primary end point was measured by the alkaline heme method to assess the reduction in severe menstrual bleeding in patients. Clinical remission was defined as a menstrual blood loss of less than 80 ml at 6 months and a reduction in blood loss from baseline to 6 months by at least 50%. Secondary endpoints included hemorrhage, hemorrhage inhibition, and hemoglobin changes. The study was conducted at approximately 100 locations in the United States and Canada.
The results of this trial showed that at the 6th month of the study, 300 mg of elagolix combined with low-dose hormonal therapy (estradiol 1.0 mg / norethisterone acetate 0.5 mg) was administered twice a day, which helped 76.2% (p < 0.001). Women with uterine fibroids reduce severe menstrual bleeding and achieve clinical remission. Elagolix's overall safety profile is consistent with that observed in the uterine fibroid phase 2 clinical trial and the first phase 3 clinical trial (ELARIS UF-I). Phase 3 research data from ELARIS UF-II will support the regulatory review submitted to elagolix. In this ongoing study, researchers will continue to collect safety data, including observations of the most common adverse events. The main results of the six-month analysis are consistent with the results of the ELARIS UF-I study reported in February 2018 and will be made public at the upcoming medical conference. Female patients in the study will continue to follow-up after treatment, or a six-month blinded extension study.
"The millions of women diagnosed with uterine fibroids currently have limited non-surgical options," said Dr. Dawn Carlson, vice president of general drug development at AbbVie. "The results of this study suggest that elagolix may have uterine fibroids. The important oral treatment options for women."
We hope that this important new uterine fibroids will pass the regulatory review and reduce the pain and recovery for the majority of female patients.
Reference materials:
[1] AbbVie's Elagolix Dazzles in Phase III Trial
[2] AbbVie Announces Positive Topline Results from Second Phase 3 Study Evaluating Investigational Elagolix in Women with Uterine Fibroids
[3] WuXi PharmaTech-Aibowei uterine fibroids new drug phase 3 clinical trial data positive
Sleep Aid Mints,Sleep Mintsid,Sleep Aid Mint,Best Sleep Aid Mints
Soar Biotech Co.,Ltd , https://www.xylitgum.com